2-Methylindole
Appearance
(Redirected from 2-methyl indole)
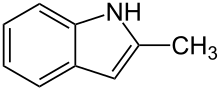
| |

| |
| Names | |
|---|---|
| Preferred IUPAC name
2-Methyl-1H-indole | |
| Identifiers | |
3D model (JSmol)
|
|
| ChEMBL | |
| ChemSpider | |
| ECHA InfoCard | 100.002.181 |
PubChem CID
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| C9H9N | |
| Molar mass | 131.178 g·mol−1 |
| Appearance | Yellow viscous liquid |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Methylketol or 2-methylindole is a mildly toxic and slightly flammable organic compound which occurs as a white solid which turns brown over time.[1][2] It has chemical formula C9H9N.[3]
Methylketol is used as an intermediate for synthesizing dyes,[4] pigments,[5] optical brighteners, and pharmaceuticals.[6]
See also
[edit]- Indole
- Methyl
- 1-Methylindole
- 5-Methylindole
- 7-Methylindole
- Skatole (3-methylindole)
References
[edit]- ^ Brown, Daniel R.; Clark, Bryan W.; Garner, Lindsey V. T.; Di Giulio, Richard T. (June 2015). "Zebrafish cardiotoxicity: the effects of CYP1A inhibition and AHR2 knockdown following exposure to weak aryl hydrocarbon receptor agonists". Environmental Science and Pollution Research International. 22 (11): 8329–8338. Bibcode:2015ESPR...22.8329B. doi:10.1007/s11356-014-3969-2. ISSN 1614-7499. PMC 4442063. PMID 25532870.
- ^ Brown, D. R.; Clark, B. W.; Garner, L. V. T.; Di Giulio, R. T. (October 2016). "Embryonic cardiotoxicity of weak aryl hydrocarbon receptor agonists and CYP1A inhibitor fluoranthene in the Atlantic killifish (Fundulus heteroclitus)". Comparative Biochemistry and Physiology. Toxicology & Pharmacology. 188: 45–51. doi:10.1016/j.cbpc.2016.05.005. ISSN 1532-0456. PMID 27211013.
- ^ Parker, Amie; Lamata, Pilar; Viguri, Fernando; Rodríguez, Ricardo; López, José A.; Lahoz, Fernando J.; García-Orduña, Pilar; Carmona, Daniel (2020-10-12). "Half-sandwich complexes of osmium containing guanidine-derived ligands". Dalton Transactions. 49 (39): 13601–13617. doi:10.1039/d0dt02713h. hdl:10261/233133. ISSN 1477-9234. PMID 32975256. S2CID 221913494.
- ^ Casas-Catalán, María José; Doménech-Carbó, María Teresa (May 2005). "Identification of natural dyes used in works of art by pyrolysis-gas chromatography/mass spectrometry combined with in situ trimethylsilylation". Analytical and Bioanalytical Chemistry. 382 (2): 259–268. doi:10.1007/s00216-005-3064-0. ISSN 1618-2642. PMID 15782338. S2CID 44407380.
- ^ Shaki, Hanieh; Gharanjig, Kamaladin; Khosravi, Alireza (July 2015). "Synthesis and investigation of antimicrobial activity and spectrophotometric and dyeing properties of some novel azo disperse dyes based on naphthalimides". Biotechnology Progress. 31 (4): 1086–1095. doi:10.1002/btpr.2107. ISSN 1520-6033. PMID 25967675. S2CID 25349059.
- ^ Ozcan-Sezer, Senem; Ince, Elif; Akdemir, Atilla; Ceylan, Özlem Öztürk; Suzen, Sibel; Gurer-Orhan, Hande (May 2019). "Aromatase inhibition by 2-methyl indole hydrazone derivatives evaluated via molecular docking and in vitro activity studies". Xenobiotica; the Fate of Foreign Compounds in Biological Systems. 49 (5): 549–556. doi:10.1080/00498254.2018.1482029. ISSN 1366-5928. PMID 29804490. S2CID 44062839.
